
Approach to a Substituted Heptamethine Cyanine Chain by the Ring Opening of Zincke Salts | Journal of the American Chemical Society
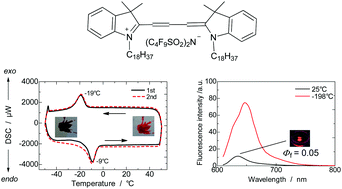
Survey, fluorescence spectra, and solubility of liquid cyanine dyes - New Journal of Chemistry (RSC Publishing)

PDF) Synthesis and absorption spectra of new polymethine cyanine dyes | Reda Abd El-Aal - Academia.edu

Synthesis of asymmetric cyanine dyes: 2, 10–13a R¹ = R² = H, R³ = C2H5;... | Download Scientific Diagram

Novel Water-Soluble Near-Infrared Cyanine Dyes: Synthesis, Spectral Properties, and Use in the Preparation of Internally Quenched Fluorescent Probes | Bioconjugate Chemistry

Explorations into the Effect of meso‐Substituents in Tricarbocyanine Dyes: A Path to Diverse Biomolecular Probes and Materials - Exner - 2021 - Angewandte Chemie - Wiley Online Library
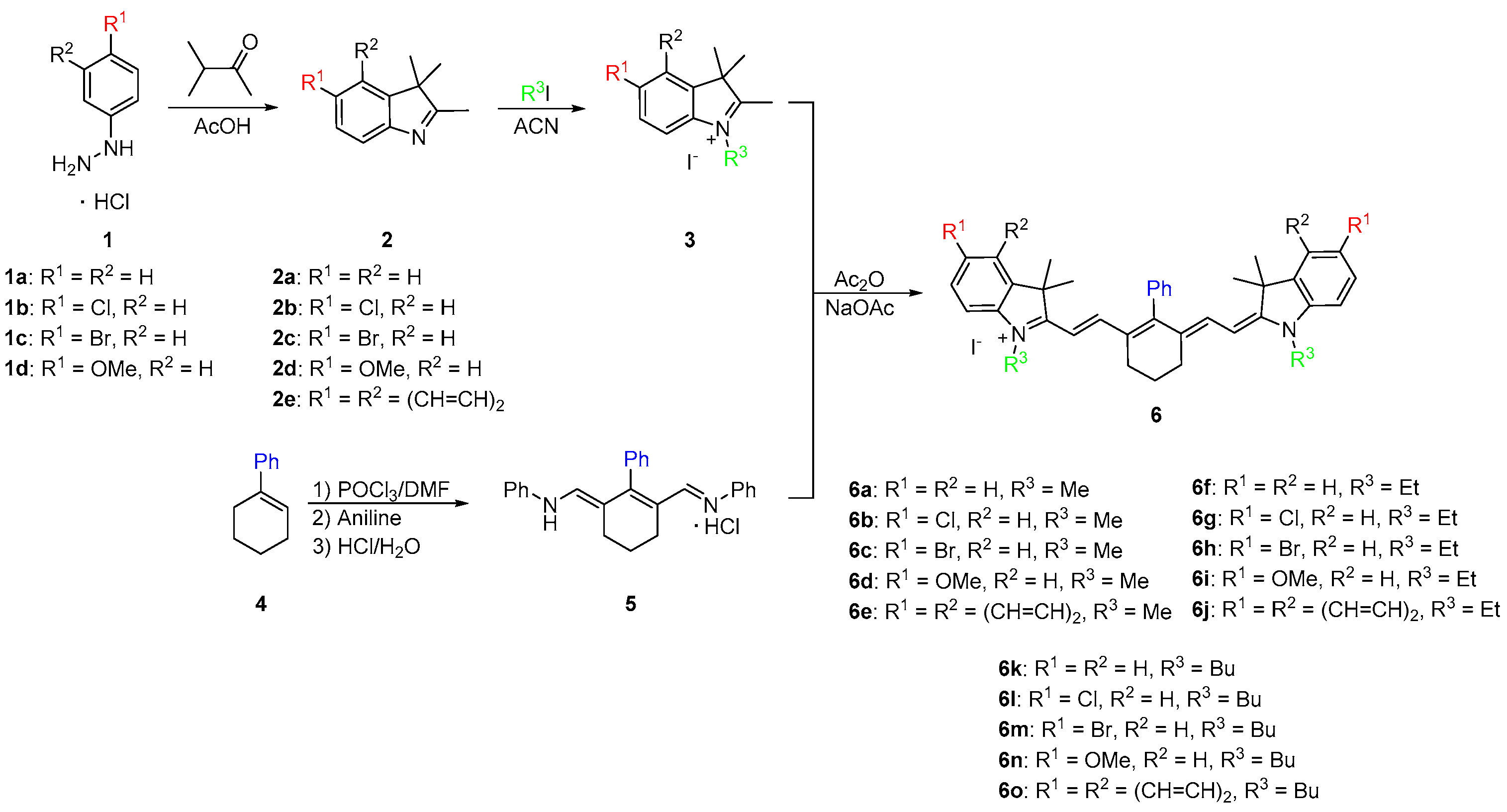
Molecules | Free Full-Text | Synthesis and Optical Properties of Near-Infrared meso-Phenyl-Substituted Symmetric Heptamethine Cyanine Dyes
A naked-eye and ratiometric near-infrared probe for palladium via modulation of a π-conjugated system of cyanines

Indolizine-Cyanine Dyes: Near Infrared Emissive Cyanine Dyes with Increased Stokes Shifts | The Journal of Organic Chemistry

Near-infrared heptamethine cyanine dyes. Synthesis, spectroscopic characterization, thermal properties and photostability - ScienceDirect

Explorations into the Effect of meso‐Substituents in Tricarbocyanine Dyes: A Path to Diverse Biomolecular Probes and Materials - Exner - 2021 - Angewandte Chemie - Wiley Online Library



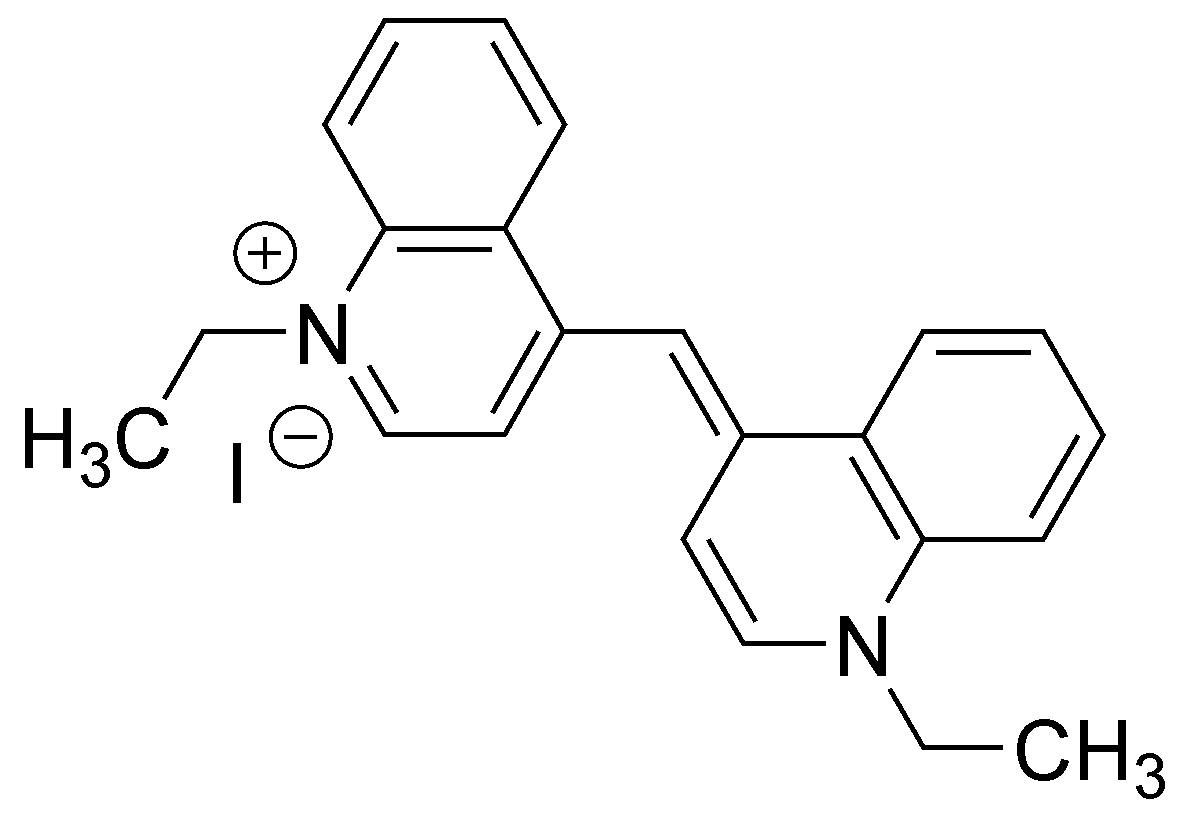

![PDF] Development of photostable near-infrared cyanine dyes. | Semantic Scholar PDF] Development of photostable near-infrared cyanine dyes. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/337f15f22c8d98132d4edbac1b79662a1df18882/2-Table1-1.png)




![PDF] Development of photostable near-infrared cyanine dyes. | Semantic Scholar PDF] Development of photostable near-infrared cyanine dyes. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/337f15f22c8d98132d4edbac1b79662a1df18882/1-Figure1-1.png)
