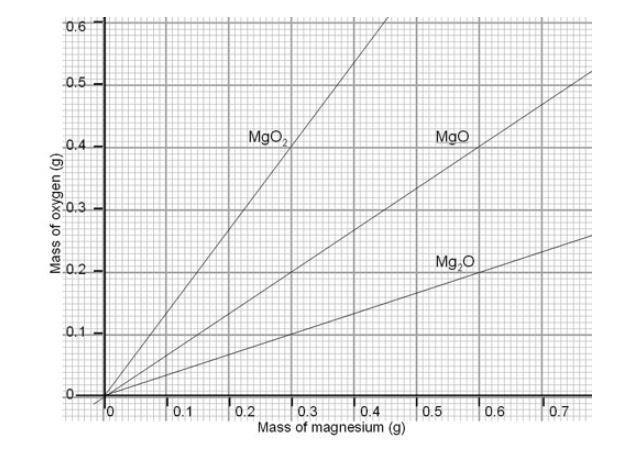
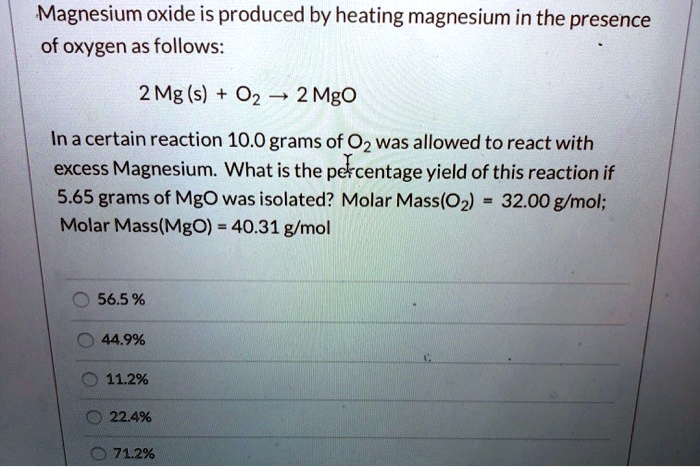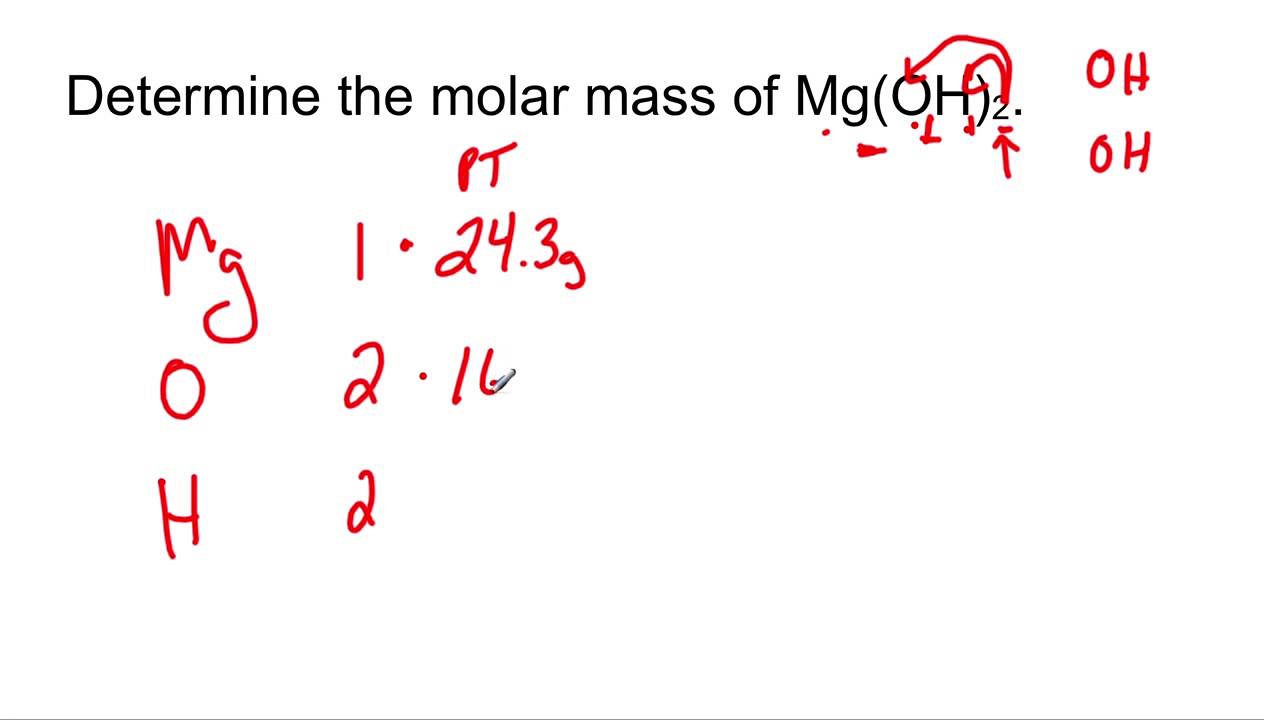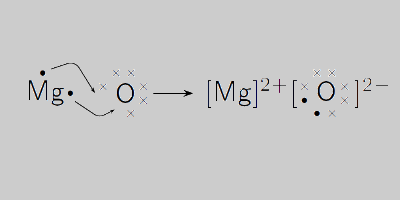![Calculate the number of molecules present in 0.5 moles of magnesium oxide ( MgO) . [Atomic weights : Mg = 24, O = 16 ] Calculate the number of molecules present in 0.5 moles of magnesium oxide ( MgO) . [Atomic weights : Mg = 24, O = 16 ]](https://toppr-doubts-media.s3.amazonaws.com/images/9085118/105391b5-2617-461a-9970-1a42367b289a.jpg)
Calculate the number of molecules present in 0.5 moles of magnesium oxide ( MgO) . [Atomic weights : Mg = 24, O = 16 ]

OB: Intro to the Mole You gotta have a reference table and a calculator of your own now. No exceptions. - ppt download

1a.calculate the relative formula mass of magnesium oxide when the relative atomic masses are O=16 - Brainly.com

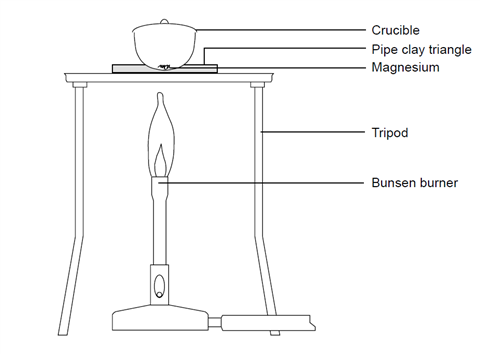
SOLVED: EXPERIMENT 4 Stoichiometry (Il: Empirical Formula of an Oxide REPORT SHEET Name: ID Mass of empty crucible weighing) b. Average mass of empty crucible Mass of crucible with Mg before heating (

What is the molar mass of magnesium oxide? I want the answer not to be round and to be used by this periodic chart - eNotes.com
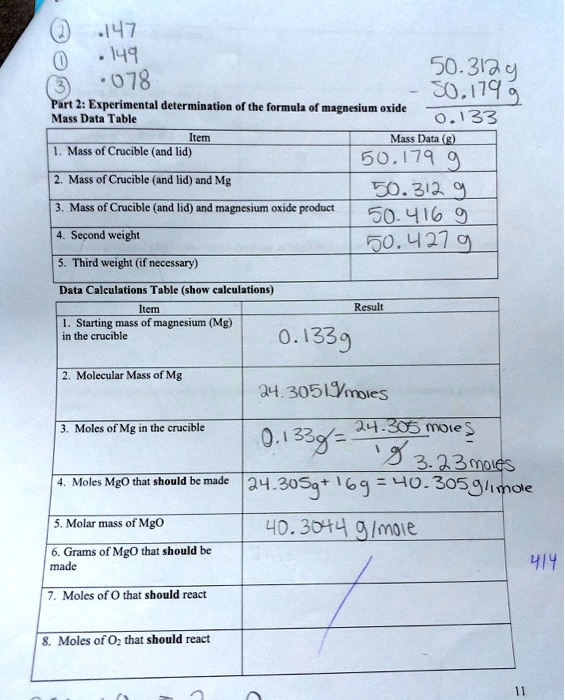
SOLVED: .147 149 50.313,9 078 50.179 Part 2: Experimental determination of the formulz of magnesium oxide Mass Data Table 0.133 Ms? Data Mass of Cricible (and lid) 50.179 Mass of Crucible (and

Objective: To reintroduce the mole and introduce two mole conversions Do Now: Solve each proportion = x2. 10 = x. - ppt download
A 3.250g sample of magnesium is burned in a container of 12.500g oxygen. What mass of oxygen gas remains unreacted after the magnesium has been completely consumed to form magnesium oxide as