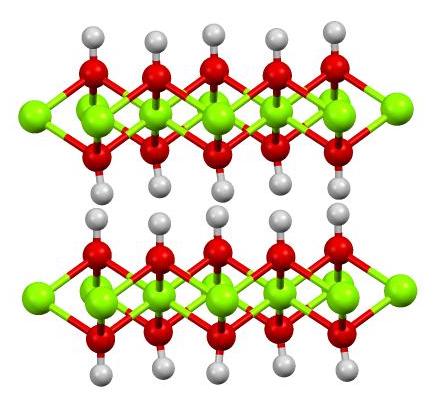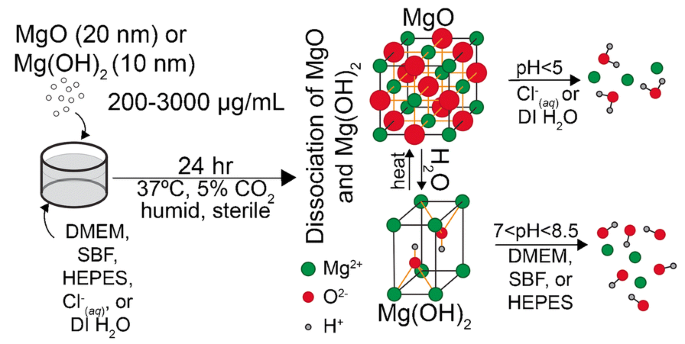
Dissociation of magnesium oxide and magnesium hydroxide nanoparticles in physiologically relevant fluids | SpringerLink

Room temperature stable COx-free H2 production from methanol with magnesium oxide nanophotocatalysts | Science Advances

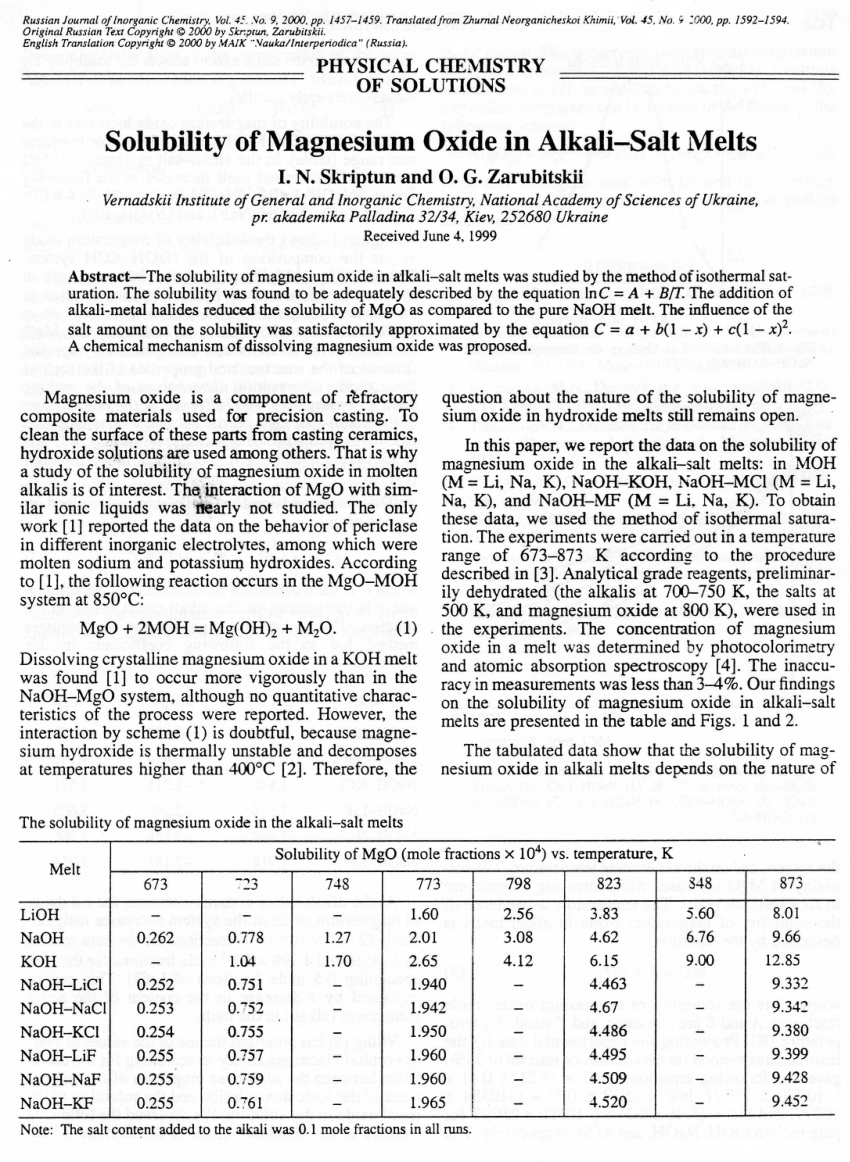
On some regularities of magnesium oxide solubility in melts with different content of alkaline earth metal chlorides - ScienceDirect

A mixture consists of four substances.The properties of each of these substances are shown in the table.Magnesium nitrate decomposes on heating to form solid magnesium oxide oxygen gas and nitrogen dioxide gas.
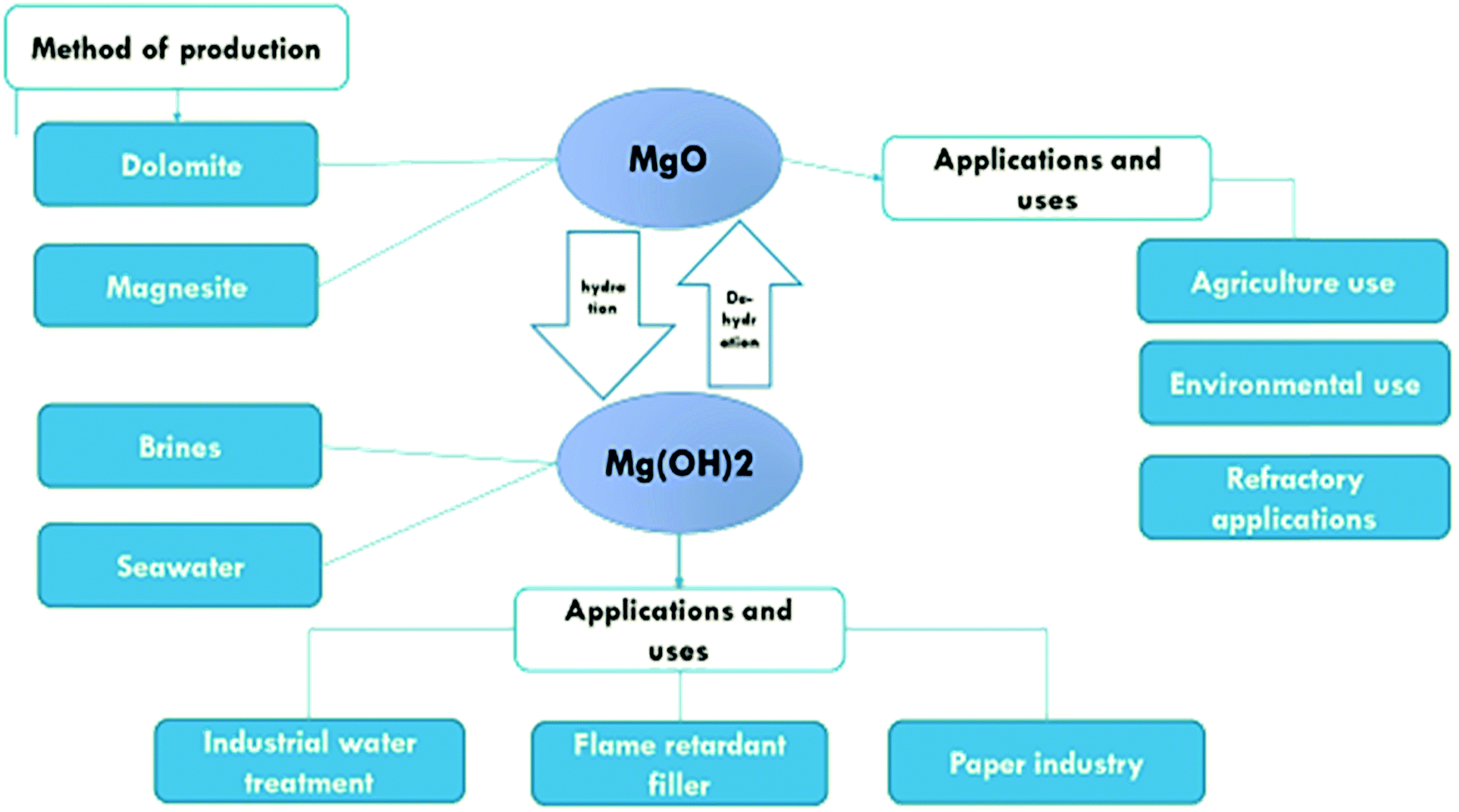
Effect of different parameters on caustic magnesia hydration and magnesium hydroxide rheology: a review - Materials Advances (RSC Publishing) DOI:10.1039/D0MA00887G
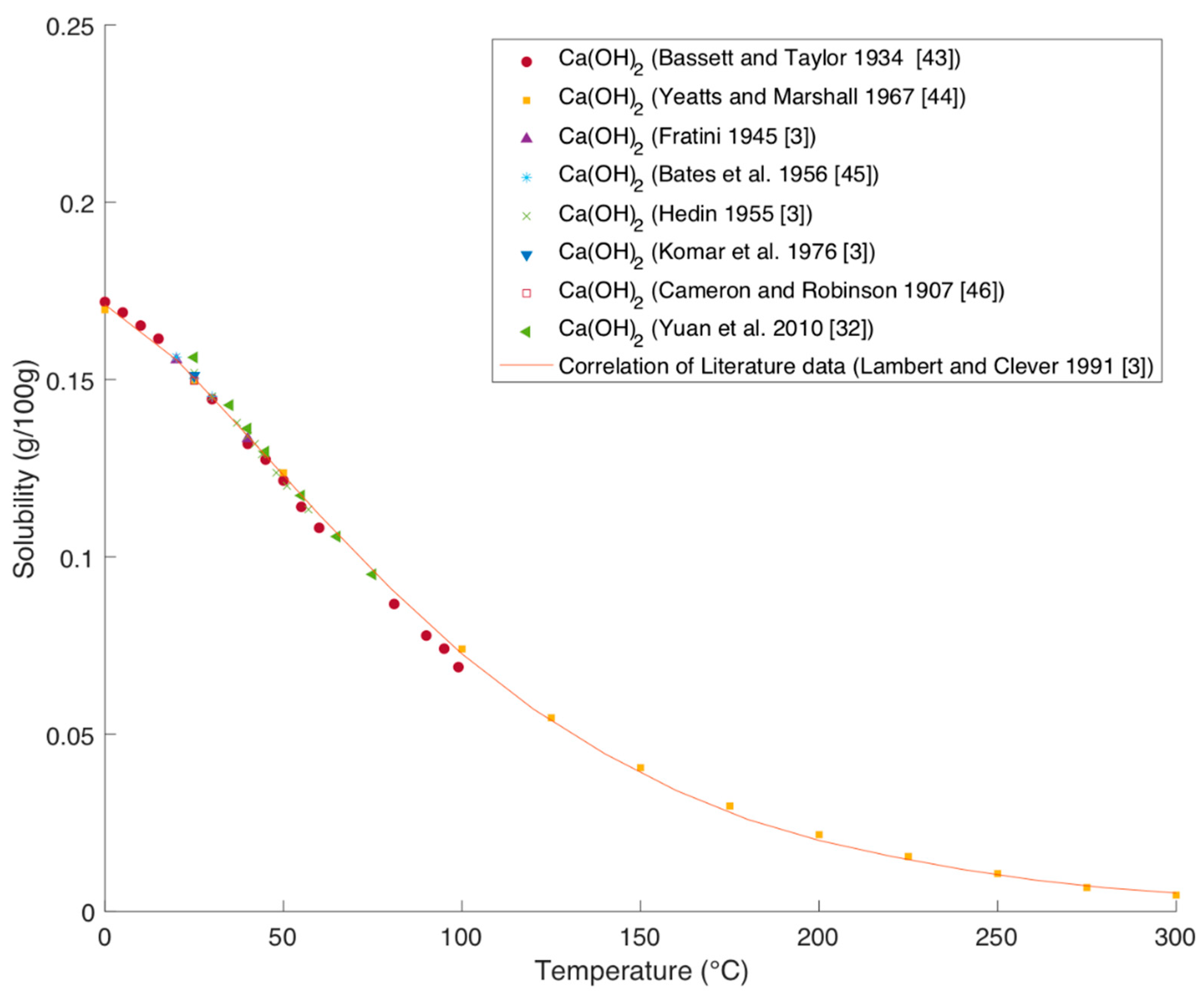
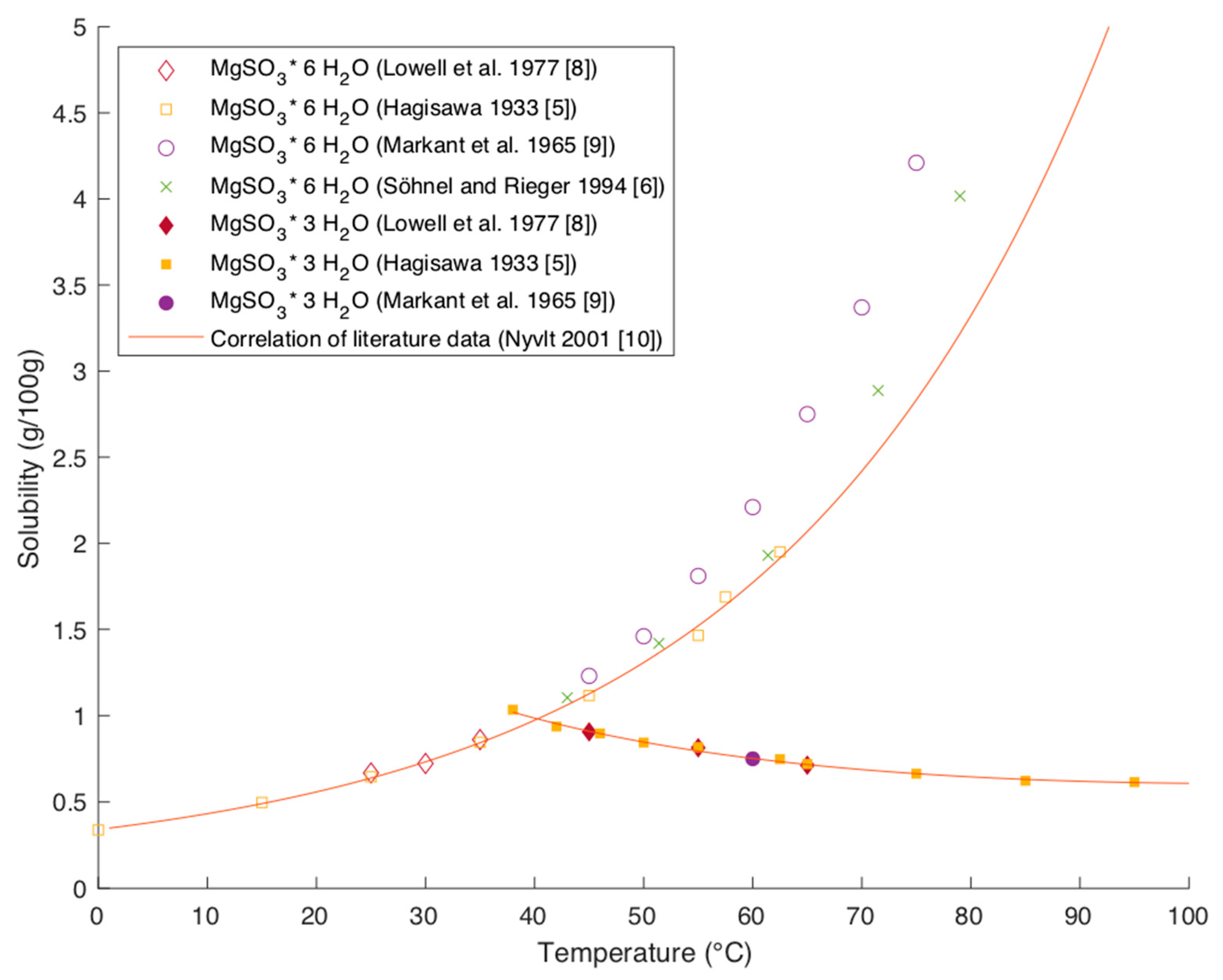
Processes | Free Full-Text | Solubility Data of Potential Salts in the MgO-CaO-SO2-H2O-O2 System for Process Modeling

Figure 1 from Magnesium bioavailability from magnesium citrate and magnesium oxide. | Semantic Scholar