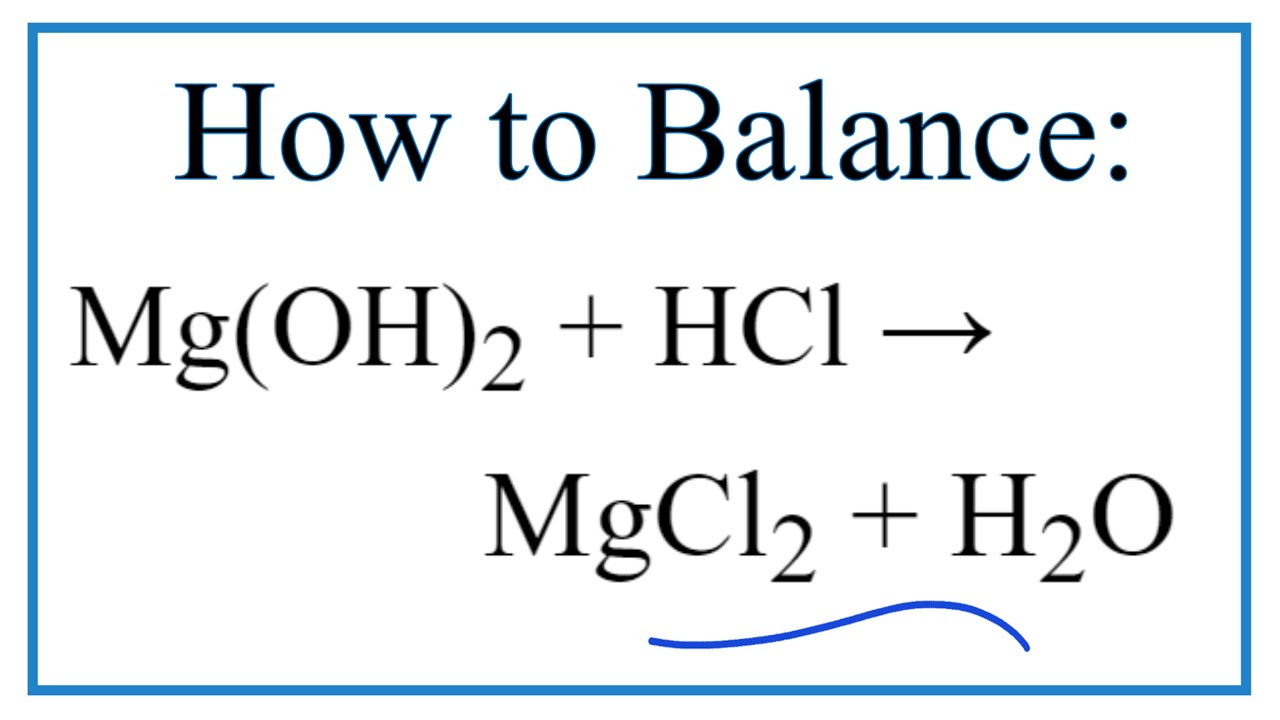
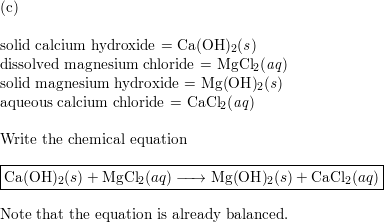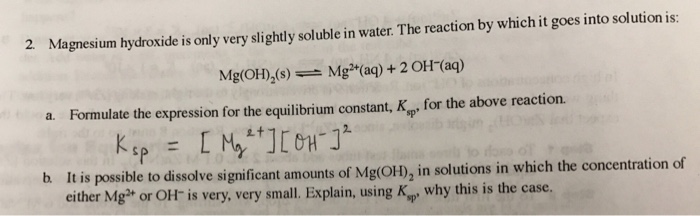SOLVED: 'Gaseous water (Hzo) and solid magnesium oxide (MgO) are formed bY the decomposition of solid magnesium hydroxide (Mg(OH)2: Write a balanced chemical equation for this reaction. D-0 Contlnue Aa 1'

Practice Write a balanced formula equation for: aluminum + oxygen aluminum oxide Al +Al OO2 →O2 →Al 2 O ppt download

why calcium and magnesium floats on water - Science - Metals and Non-metals - 13061069 | Meritnation.com

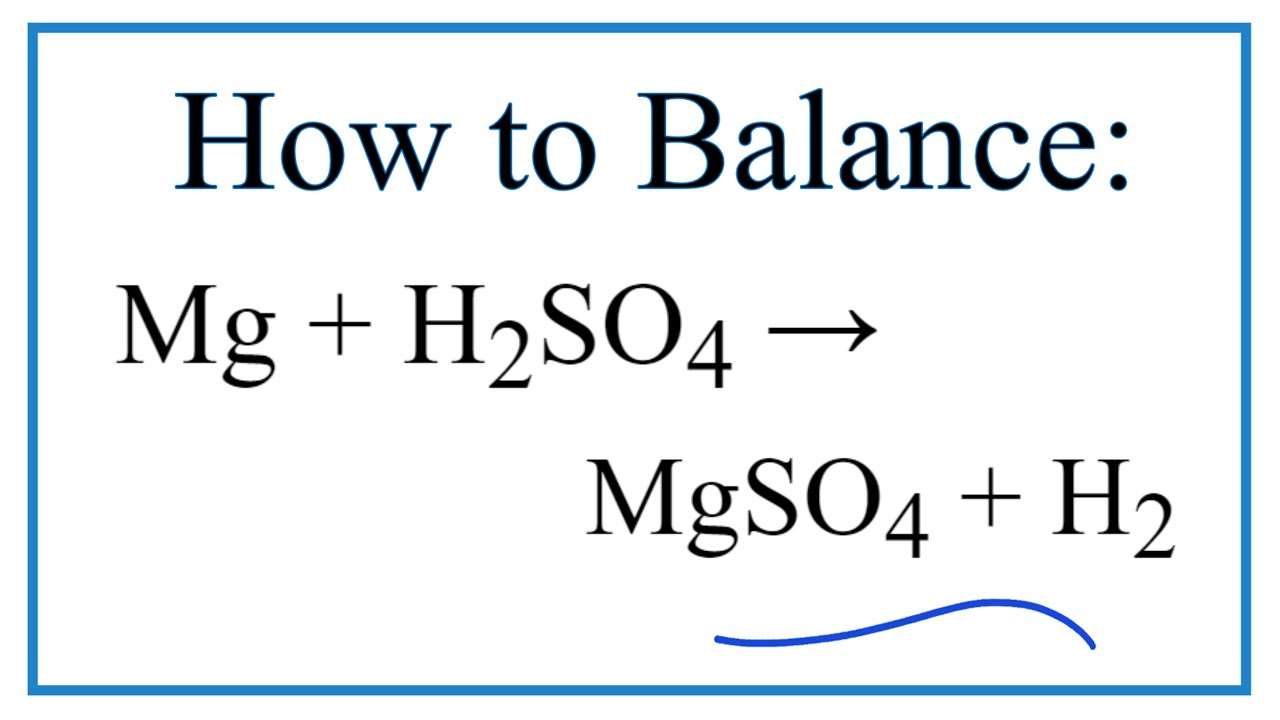
SOLVED:Write balanced equations for each reaction. (Chapter 9) a. Magnesium metal and water combine to form solid magnesium hydroxide and hydrogen gas. b. Dinitrogen tetroxide gas decomposes into nitrogen dioxide gas. c.